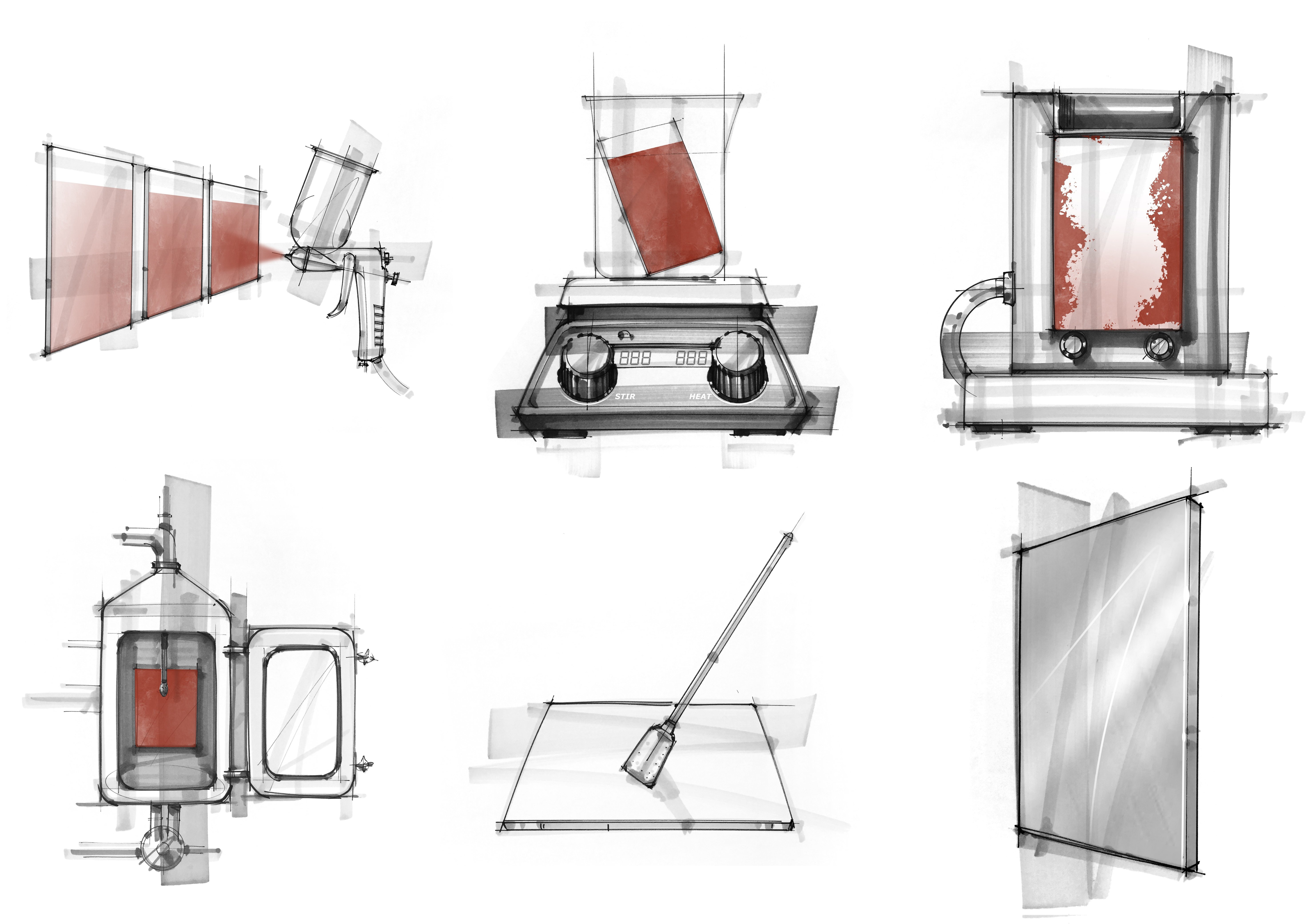
Failures in Equipment Cleaning & Criteria for Passing Results: Lab Scale
In the Dober Chematic Lab, we aim to make a difference and use our years of cleaning experience to help determine what clean really is.
After working with many soil types, equipment, and capabilities, we have seen some interesting failures in the lab-scale cleaning process. From these failures, we have created a list of criteria for how we determine a passing result.
Section 1: Visible Residue
Out of all the evaluation criteria, this can be the most straightforward.
Visible residue can be left on the coupon surface in many ways. Some soils leave behind a large amount of bulk. When looking at bulk soil that is left, it may be the whole soil that was not removed effectively, or it may be a layer of soil from a multilayer application.
There are also situations where the visible residue left behind is not as easily seen. However, any trace left behind is considered an ineffective clean and more work needs to be completed.
Any visible residue, no matter how small, can cause larger problems in the future.



Section 2: Water Break
Evaluating to make sure that there is a “water break free” surface is recognized by American Society for Testing and Materials under ASTM F22. This method helps us recognize if there are hydrophobic materials left on the surface we are evaluating.
A water break can be observed to occur quickly after a water rinse is finished. If the coupon is observed closely after the rinse step, a water break may be missed. The importance of catching a water break is that for many of these instances, a coupon that only has an observable water break may have no other sign of failure. This, in turn, over time can cause subsequent failures and, potentially, a visible residue to appear.
Our rule of thumb is that the surface should have an even sheet of water on the surface for at least 10 seconds, then there is a significant chance that there will be no sign of a water break.



Different types of water break that can be observed based on soil type, process or incorrect cleaning agent versus a water break-free surface (image on the far right).
Section 3: Hydrophilic Gel Layers
Products that cause hydrophilic gel layers can contain ingredients such as polymers, starches, or sugars that have a strong affinity or absorption for water.
This absorption of water causes the soil on the coupon to swell and cling onto the coupon surface, causing it to be increasingly difficult to remove from the surface.
Most gel layers are hard to observe in general, let alone in lab scale. When a gel layer is missed with the initial cleaning process, we typically see cleaning issues with subsequent batches as raw materials are trapped in the gel layer and become difficult to remove. Most gel layers appear as a small, swelled line, usually at the edge of where a soil line and the rest of the coupon meet. Other times, the whole product swells and then dries on the coupon surface.
From our experiences with different soils, gel layers can be observed in two main ways (as displayed below).

 The picture on the left is the most common and hard to observe, only showing a swelled line on part of the coupon. The less common but easier to see is the photo on the right. Some products, once the coupon is dry, leave what we call “a tan haze."
The picture on the left is the most common and hard to observe, only showing a swelled line on part of the coupon. The less common but easier to see is the photo on the right. Some products, once the coupon is dry, leave what we call “a tan haze."
Section 4: Hidden Residues – Differing Light Sources
Light sources are a helpful trick when it comes to bringing soil and residues to center stage.
The light source that is used to view equipment, large or small scale, can occasionally make a difference in whether or not you catch leftover soil or residue on your equipment. Most times, normal laboratory or warehouse lighting is the only resource facilities may have to view surfaces. However, this type of lighting does not always show the fine details. We have found that having a second light source can be an easy and effective way to find failures in cleaning.
In our lab scale studies, we observe all coupons under a 50W halogen light source. This is a resource that is inexpensive and small but critical. The best part about this is that it can also be utilized in large-scale studies, as well, with a stand light that can investigate larger pieces of equipment.




Regular Lighting 50W Halogen Lighting
Section 5: Consistent Cleaning by Replication
The final and most important way to determine a passing result in lab scale is cleaning replication.
Once effective cleaning parameters and cleaning agent are determined, the next objective is to make sure the process can be repeated. It is crucial to prove that a cleaning process can be performed more than once, as that is expected in facilities when scaled up.
In our laboratory-scale studies, we repeat all chosen cleaning methods in triplicate to ensure a consistent clean before we share the results with customers. This ensures that the cleaning agent and chosen parameters will be a robust and reliable recommendation time after time, giving customers one less thing to worry about.
There are many small but mighty criteria that can be performed in a lab-scale setting that can help guarantee scaling up will be a breeze. Small details can end up being large issues, so it is critical to catch every possible roadblock before it occurs. Fixing issues in the small scale helps save time and money in the large scale.
Have any questions? Reach out to us and a Chematic cleaning expert will get back to you.