Wastewater Coagulation
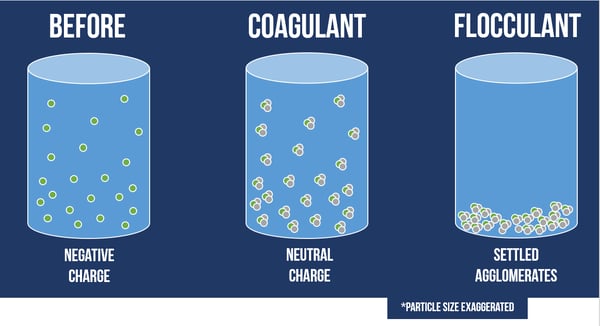
Wastewater coagulation is the chemical water treatment process used to remove solids from water, by manipulating electrostatic charges of particles suspended in water.
This coagulation process introduces small, highly charged molecules into water to destabilize the charges on particles, colloids, or oily materials in suspension.
Selecting the right coagulant for a system will enhance overall system performance, and particularly improve solids removal efficiency by enhancing filter and clarifier performance.
Wastewater Coagulation Applications
There are many wastewater treatment applications that require coagulation reactions, such as removing colloidal solids from water, demulsifying oil emulsions (“emulsion breaking”), and in paint detackification. There are also many types of coagulants available to meet specific needs of a treatment process. In general, coagulation precedes flocculation in a chemical water treatment process.
Wastewater Coagulation Particles: Types and Settling Times
Particles in water carry an electrostatic charge on their surface. Common examples include clay, silica, iron, paints, and even oil. These small, suspended particles are stabilized in suspension & difficult to remove via mechanical methods.
A suspension of solids in water typically contains a variety of particles sizes. A lab analysis of “particle size distribution” will help define the size of particles as well as the relative amount of each size particle in the suspension.
Particles over 100μm are generally considered “settleable solids” and readily settle out of suspension. Particles sized 10-100μm are generally considered “turbidity” and are often addressed in a wastewater treatment system with coagulation. Particles smaller than 10μm are “colloidal particles”, which are almost always treated with coagulation because it is quite expensive to remove small particles using only mechanical water treatment like filtration.
|
Diameter of Particle |
Type of Particle |
Settling Time in 1m Water |
|
|
10mm |
Gravel |
1 second |
Settleable Solids |
|
1mm |
Sand |
10 seconds |
|
|
100µm |
Fine Sand |
2 minutes |
Turbidity |
|
10µm |
Silt, Dust |
2 hours |
|
|
1µm |
Clay |
8 days |
Colloidal Solids |
|
0.1µm |
Colloids |
2 years |
Colloidal particles are further classified as hydrophobic and hydrophilic colloids. Their respective “water-hating” or “water-loving” nature is important in wastewater treatment. The hydrophobic colloids do not chemically react with a coagulant, while hydrophilic colloids may chemically react with a coagulant used in the treatment process. As a result, hydrophilic colloids, such as dyes, require more coagulant than hydrophobic colloids.
Electrostatic charges of particles in water work with the familiar statement about magnetics, “Like repels like and opposites attract." The terms used to describe charges are “Cationic” which refers to a positive charge and “Anionic” which refers to a negative charge. Due to the chemistry of water, most particles carry a negative charge.
In addition to the positive or negative character of a charge, the strength of that electrostatic charge is referred to as “Zeta potential." The strength of a charge is very important in wastewater treatment because stronger charges create more stable suspension of particle in water. Zeta potential is measured on a scale of -61 → +61, where further from 0 is a stronger negative or positive charge with a more stable suspension in water. Near 0, particles will readily fall out of suspension, while escalating much beyond ±10 will require coagulation.
|
Zeta potential (mV) |
Particle stability in suspension |
|
0 to ±5 |
Rapid coagulation or flocculation |
|
±10 to ±30 |
Early instability |
|
±30 to ±40 |
Moderate stability |
|
±40 to ±60 |
Good stability |
In the context of most chemical water treatment processes, coagulation should occur upstream of flocculation. With coagulation, destabilized particles begin to collide and create small masses, often called “pin flocs” or “micro flocs," since they are barely visible to the naked eye at around 50 μm in size. Flocculation is the process of clumping particles together to build larger agglomerates. This process introduces a large molecule with electrostatically charged binding sites to attract oppositely-charged particles or microflocs. The flocculation reaction itself is highly visible, as the resulting “flocs” readily separate from the water.
Do note, the terms “coagulation” and “flocculation” are often used interchangeably, but they are in fact distinct functions.
Wastewater coagulation products
There are many coagulants available for wastewater treatment. Below is an introduction to the coagulants representing the variety of different formulations and charge densities.
Metals-based coagulants form the largest group of products available. Products may contain only metal salt (e.g., aluminum sulfate or ferric chloride), or products may be polymerized metal salts (e.g., polyaluminum chloride or aluminum chlorohydrate).
Synthetic coagulants may carry very high charge densities on relatively large molecules (such as polyamine or polyDADMAC). Depending on the formulation, some of the synthetic derivatives may behave as a flocculant.
Biopolymer coagulants are derived from natural sources. A number of Dober Water Treatment products are derived from plant sources (like lignin, tannins and starches). There are also polysaccharides and other natural biopolymers that are sourced from animals, fungi and microbial processes (to generate chitosan, pectin, carrageenan, plantago, etc.).
For these sources and formulations of coagulants, many hybrids exist. By combining two or more coagulants, even more options are available.
Dober's water treatment division specializes in leveraging the best characteristics of coagulants to create hybrids that optimize a variety of wastewater treatment processes.
It is possible to test at the bench-scale how a coagulant will perform in a wastewater treatment process. Jar testing is an essential tool for comparing products, optimizing performance, estimating treatment costs and planning operations.
Regardless of which coagulant chemistry is integrated in a wastewater treatment train, coagulation reactions require adequate mixing. Typical options to accomplish mixing are an in-line static mixer or a coagulation tank with a powered mixer.
Equipment selection will depend on the overall system design and operation, but the goal remains the same. Coagulant molecules need to interact and collide with particles in suspension to break apart the colloidal system.
Historically, metal salt-only coagulation (alum, ferric chloride, etc.) can create excess sludge for disposal or create safety and toxicity issues downstream of the treatment discharge. Effective coagulation can help stabilize treatment performance through the variability of industrial operations with product changeovers, sanitation activities and flow changes.
Selecting the right coagulation step will enhance the entire treatment system with more efficient solids separation.
Download Your Copy of Our Product Catalog



